This is the one that sounds like a stretch until you understand the mechanism.
Gut health and hyperpigmentation do not seem like they should be connected. One lives in your digestive system. The other lives on your face. But your skin does not operate independently from the rest of your body. It depends on an internal supply chain: nutrients absorbed through the gut, inflammatory signals regulated by gut function, and systemic conditions shaped by what is happening in your digestive system long before anything reaches the surface.
When that supply chain is working, your skin gets what it needs to regulate melanocyte activity, turn over pigmented cells, repair damage, and manage inflammation. When it is compromised, the skin does not stop working. It just works worse. Pigment regulation becomes less precise. Repair slows down. The inflammatory environment shifts toward conditions that favour pigmentation.
This is not about superfoods or detox teas. It is about the basic biological infrastructure your skin depends on and what happens when that infrastructure is quietly failing.
The gut-skin connection is not abstract
Your gut does two things that directly affect your skin's capacity to manage pigment.
The first is absorption. Your gut is where nutrients from food enter your bloodstream and become available to the rest of your body, including your skin. Every enzyme involved in melanin production, every antioxidant that protects skin cells from oxidative damage, every cofactor required for cell turnover depends on nutrients that were absorbed through the gut lining. If absorption is impaired, the skin does not get what it needs, regardless of what you eat.
The second is immune regulation. A significant portion of your immune system operates in and around the gut. The gut microbiome (the community of bacteria living in your digestive tract) plays a direct role in calibrating systemic inflammatory signalling. When the microbiome is disrupted, or when the gut lining becomes more permeable than it should be, inflammatory signals increase throughout the body, not just in the gut.
Both pathways converge on the same outcome: a skin that is less equipped to regulate pigment and more susceptible to the triggers that produce it.
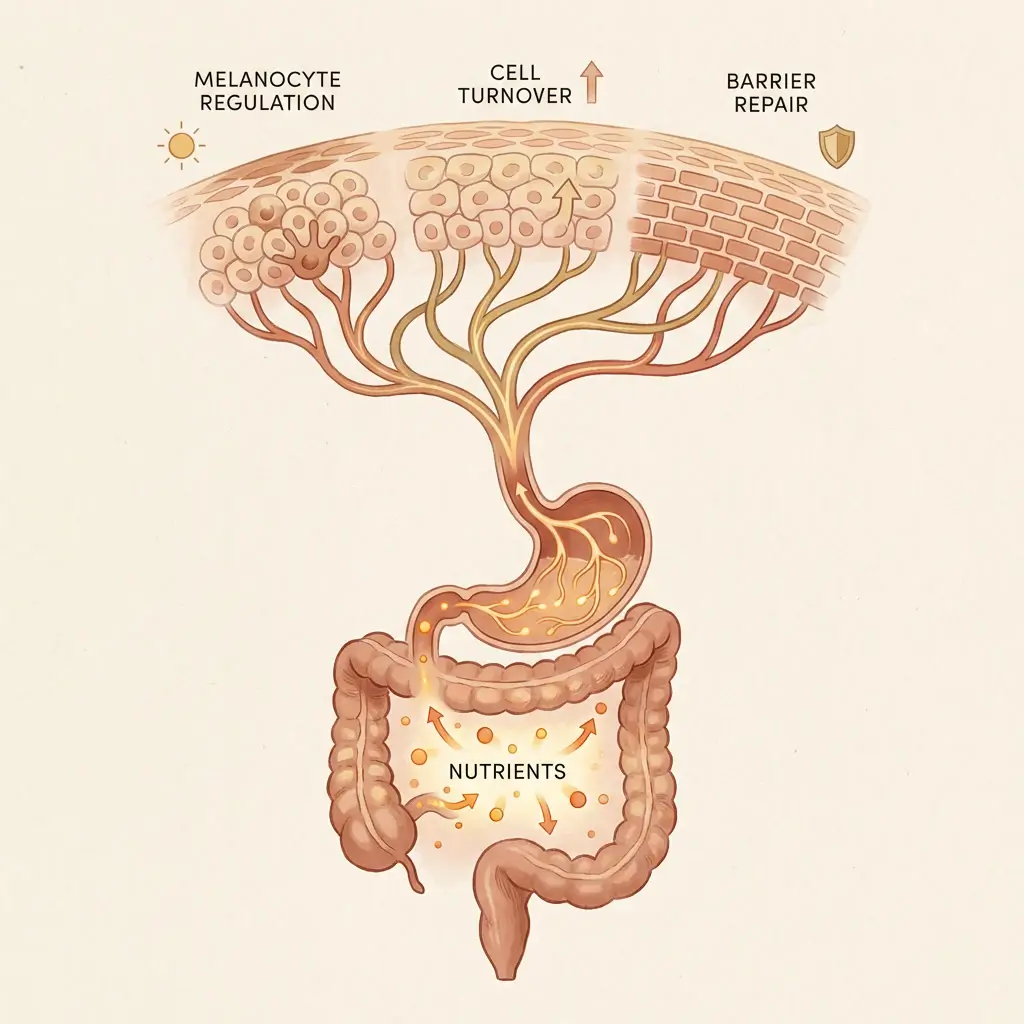
How gut dysfunction creates the conditions for pigmentation
Gut dysfunction is a broad term, and it does not mean a dramatic diagnosis. It can be subtle and chronic, the kind of thing that produces vague symptoms (bloating, irregular digestion, food sensitivities) without ever crossing a clinical threshold. But even at mild levels, it affects the internal environment your skin operates in.
Poor nutrient absorption
Conditions that affect the gut lining (chronic inflammation, coeliac disease, irritable bowel conditions, long-term use of certain medications, even sustained low-grade dysbiosis) can reduce the gut's ability to absorb nutrients efficiently. You can be eating well and still be functionally deficient in the micronutrients your skin depends on, because the absorption step is compromised.
This matters for pigmentation because melanocyte regulation, antioxidant defence, and cell turnover are all nutrient-dependent processes. They do not run on willpower. They run on raw materials. When the raw materials are not arriving, the processes degrade quietly. The skin does not send an obvious distress signal. It just becomes less capable of doing what it needs to do, and pigmentation is one of the places that shows up.
Increased gut permeability
When the gut lining becomes more permeable than it should be (sometimes called intestinal hyperpermeability), molecules that would normally be contained within the digestive tract enter the bloodstream. The immune system recognises these as foreign and mounts an inflammatory response.
This is not a one-time event. If the permeability is ongoing, the inflammatory signalling is ongoing. The result is a chronic elevation in systemic inflammation that your melanocytes sit in constantly. The same ambient inflammatory environment that chronic stress or blood sugar dysregulation creates. The source is different. The downstream effect on melanocyte sensitivity is similar.
Microbiome disruption
The gut microbiome influences systemic inflammation through multiple pathways. A balanced, diverse microbiome helps calibrate immune responses and keep inflammatory signalling in check. When that balance is disrupted (by antibiotics, poor diet, chronic stress, or other factors), the regulatory function weakens and inflammatory tone increases.
Research on the gut microbiome and skin pigmentation specifically is still early. But the connection between dysbiosis and systemic inflammation is well established, and systemic inflammation is one of the most consistent drivers of melanocyte reactivity. The pathway is indirect but real.
How specific nutrient deficiencies affect pigmentation
Not every nutrient matters equally for pigmentation. A handful of specific deficiencies have documented connections to melanocyte behaviour, skin repair, or the inflammatory environment that influences both.
Iron
Iron deficiency is one of the most common nutritional deficiencies worldwide, particularly in women of reproductive age. It affects oxygen delivery to tissues, including the skin. It impairs cell turnover. And it is associated with both diffuse skin pallor and, in some presentations, paradoxical hyperpigmentation, particularly in darker skin tones where the interaction between iron status and melanocyte activity is more complex.
Iron deficiency also increases oxidative stress (the body's iron-dependent antioxidant systems cannot function properly) and can worsen the fatigue and stress reactivity that compound pigmentation through other pathways.
B vitamins
Several B vitamins (particularly B12, folate, and B6) are involved in the methylation and cellular repair processes that support healthy skin turnover. Deficiency in B12 specifically has been associated with hyperpigmentation, particularly diffuse darkening of the skin, nails, and oral mucosa. This presentation is more commonly documented in melanin-rich skin.
Folate deficiency impairs DNA synthesis and cell division, slowing the turnover that clears pigmented cells from the surface. B6 is involved in the synthesis of neurotransmitters and hormones that indirectly affect inflammatory tone.
Zinc
Zinc is a cofactor in hundreds of enzymatic reactions, including those involved in immune regulation, wound healing, and antioxidant defence. Zinc deficiency impairs barrier function, increases susceptibility to inflammation, and slows the skin's repair capacity. It does not directly drive melanocyte activity, but it degrades the environment melanocytes operate in and weakens the systems that would otherwise keep pigmentation in check.
Vitamin D
The relationship between vitamin D and pigmentation is complex. Vitamin D is synthesised in the skin through UV exposure, and melanin reduces the efficiency of that synthesis. This means women with melanin-rich skin are at significantly higher risk of vitamin D deficiency, particularly in less sunny climates.
Vitamin D plays a regulatory role in immune function, inflammation, and cell differentiation. Deficiency is associated with increased inflammatory signalling and impaired skin barrier function. It does not directly trigger hyperpigmentation, but it contributes to the systemic conditions that make pigmentation more likely and harder to resolve.
Why this is not a cosmetic problem
This is the distinction that matters most. Gut dysfunction and nutrient deficiency are not skincare issues. They are health issues that show up in the skin.
You cannot fix impaired nutrient absorption with a better serum. You cannot compensate for chronic gut-driven inflammation with a stronger retinoid. These are systemic conditions that require systemic assessment. A GP, a dermatologist who thinks beyond the surface, or a practitioner who understands the gut-skin axis.
The purpose of here is not to give you a protocol. It is to help you recognise that if your pigmentation is not responding the way it should despite a well-constructed routine and adequate sun protection, the problem may not be on your skin at all. It may be in the internal systems that your skin depends on.
This is especially relevant for women who are also experiencing:
- Chronic bloating, irregular digestion, or food sensitivities
- Fatigue that does not improve with rest
- Hair thinning or brittle nails alongside skin changes
- History of restrictive eating, dieting, or prolonged dietary limitation
- Long-term use of medications that affect gut function or nutrient absorption (PPIs, metformin, certain antibiotics)
- Diagnosed conditions affecting the gut (IBS, coeliac disease, inflammatory bowel disease)
None of these prove that gut health or nutrient deficiency is driving your pigmentation. But when they appear alongside pigmentation that is not responding to standard treatment, they are worth investigating.
Why melanin-rich skin is more vulnerable to these patterns
Two factors compound the issue for women with deeper skin tones.
The first is melanocyte sensitivity. Melanocytes in melanin-rich skin are more responsive to the inflammatory shifts that gut dysfunction and nutrient deficiency create. The same ambient elevation in systemic inflammation that might produce no visible changes in lighter skin can produce noticeable pigmentation in deeper tones.
The second is vitamin D. Melanin reduces the skin's capacity to synthesise vitamin D from UV exposure. Women with melanin-rich skin are significantly more likely to be vitamin D deficient, particularly in temperate or northern climates, and that deficiency feeds directly into the inflammatory and immune dysregulation that affects pigmentation.
These are not marginal factors. They are common, clinically documented patterns that commonly affect the same population most likely to struggle with hyperpigmentation.
What to take from this
Gut health and nutrient status are not the most obvious places to look when your pigmentation will not clear. But they are among the most common internal factors quietly shaping the environment your skin operates in.
The mechanism is not exotic. Your skin needs specific nutrients to regulate melanocyte activity, turn over pigmented cells, defend against oxidative stress, and manage inflammation. Those nutrients come through the gut. When the gut is not functioning well, or when key nutrients are missing, every one of those processes degrades. Not dramatically. Not obviously. Just enough that pigmentation becomes easier to trigger and harder to resolve.
This isn't a call to self-diagnose or self-supplement. It is a call to expand the frame. If your pigmentation is resistant to treatment and you have any of the co-occurring patterns described here, a conversation with a healthcare provider about gut function and nutrient status is a reasonable and underutilised step.
Sometimes the most important thing you can do for your skin isn't something you do to your skin.






